Prev Page--Recovery, Utilization || Next Page--Formations
Ground Water, continued
Chemical Character of Ground Water
The chemical character of the ground waters in Kiowa County is shown- by the analyses of water from 27 representative wells and 5 springs given in Tables 4 and 5, respectively. The analyses, which were made by Howard Stoltenberg in the Water and Sewage Laboratory of the Kansas State Board of Health, show only the dissolved mineral content of the waters and do not in general indicate the sanitary condition of the waters. The constituents given were determined by the methods used by the U. S. Geological Survey.
Table 4--Analyses of waters from typical wells in Kiowa County, Kansas. Analyzed by H. A. Stoltenberg. Parts per milliona and equivalents per millionb (in italics).
| No. on plate 2 |
Location | Geologic subdivision | Depth (feet) |
Date of collection, 1941 |
Temp. (F°) |
Iron (Fe) |
Calcium (Ca) |
Magnesium (Mg) |
Sodium and potassium (Na+K)c |
Bicar- bonate (HCO3) |
Sulfate (SO4) |
Chloride (Cl) |
Fluoride (Fl) |
Nitrate (NO3) |
Dissolved solids |
Hardness (calculated as CaCO3) |
||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total | Carbonate | Non- carbonate |
||||||||||||||||
| T. 27 S., R. 16 W. | ||||||||||||||||||
| 3 | SE NE sec. 6 | 38 | Meade | Sept. 19 | 60 | 0.21 | 40 2.00 | 6.5 .53 | 1.2 .05 | 129 2.12 | 4.5 .09 | 4.2 .12 | 0.2 .01 | 15 .24 | 136 | 126 | 106 | 20 |
| T. 27 S., R. 17 W. | ||||||||||||||||||
| 8 | SW SW sec. 8 | 30 | Meade | Sept. 19 | 61 | .54 | 37 1.85 | 5.2 .43 | 4.6 .20 | 110 1.80 | 8.2 .17 | 2.5 .07 | .3 .02 | 26 .42 | 139 | 114 | 90 | 24 |
| T. 27 S., R. 18 W. | ||||||||||||||||||
| 13 | SE cor. SW sec. 8 | 43.5 | Meade | Sept. 19 | 61 | .86 | 62 3.09 | 6.3 .52 | 4.8 .21 | 207 3.39 | 1.6 .03 | 4.5 .13 | .2 .01 | 16 .26 | 200 | 180 | 170 | 13 |
| T. 27 S., R. 19 W. | ||||||||||||||||||
| 18 | SE SW sec. 30 | 69.5 | Meade | Sept. 19 | 60 | .5 | 56 2.79 | 7.6 .62 | 8.5 .37 | 192 3.15 | 7.8 .16 | 5 .14 | .4 .02 | 19 .31 | 201 | 170 | 158 | 12 |
| T. 27 S., R. 20 W. | ||||||||||||||||||
| 19 | SE NE sec. 1 | 46 | Meade | Sept. 19 | 61 | .04 | 25 1.25 | 5.6 .46 | 5.1 .22 | 79 1.30 | 20 .42 | 5 .14 | .2 .01 | 3.9 .06 | 104 | 86 | 65 | 21 |
| T. 28 S., R. 16 W. | ||||||||||||||||||
| 24 | SW SW sec. 8 | 160 | Meade | Feb. | .01 | 50 2.50 | 5.7 .47 | 9.6 .45 | 175 2.87 | 3.7 .08 | 10 .28 | .1 .01 | 11 .18 | 224 | 150 | 144 | 6 | |
| T. 28 R. 17 W. | ||||||||||||||||||
| 27 | SW SE sec. 8 | 100 | Meade | Sept. 20 | 61 | 1.2 | 46 2.30 | 5.6 .46 | 13 .57 | 169 2.77 | 7.6 .16 | 7 .20 | .3 .02 | 11 .18 | 176 | 138 | 138 | 0 |
| T. 28 S., R. 18 W. | ||||||||||||||||||
| 31 | SW NE sec. 16 | 125-250 | Meade and Ogallala (Composite sample from wells 31-34) | Dec. | 0 | 60 2.99 | 6.8 .56 | 18 .78 | 211 3.46 | 19 .4 | 11 .31 | .2 .01 | 9.3 .15 | 267 | 178 | 173 | 5 | |
| T. 28 S., R. 19 W. | ||||||||||||||||||
| 44 | SW NW sec. 22 | 107.5 | Meade | Sept. 19 | 61 | 1.2 | 80 3.99 | 11 .90 | 11 .49 | 267 4.38 | 16 .33 | 20 .56 | .2 .01 | 6.2 .1 | 279 | 244 | 219 | 25 |
| T. 28 S., R. 20 W. | ||||||||||||||||||
| 49 | SE SW sec. 23 | 83.8 | Meade and/or Ogallala | Sept. 19 | 59 | 4.8 | 72 3.59 | 12 .99 | 13 .57 | 251 4.12 | 21 .44 | 14 .39 | .3 .02 | 11 .18 | 274 | 229 | 206 | 23 |
| T. 29 S., R. 16 W. | ||||||||||||||||||
| 55 | NW NE sec. 33 | 100 | Flowerpot | Oct. 1 | 60 | .91 | 71 3.54 | 32 2.63 | 1,548 67.32 | 210d 3.44 | 1,722 35.88 | 1,210 34.12 | 1.9 .1 | .71 .01 | 4692 | 308 | 172 | 136 |
| T. 29 S., R. 17 W. | ||||||||||||||||||
| 56 | NW NE sec. 5 | 130 | Meade | Sept. 19 | 60 | .08 | 74 3.69 | 11 .9 | 13 .58 | 281 4.61 | 9.6 .2 | 9.5 .27 | .2 .01 | 4.9 .08 | 263 | 230 | 230 | 0 |
| 58 | NW NE sec. 23 | 40 | (?) | Oct. 1 | 60 | 5.6 | 83 4.14 | 9.6 .79 | 11 .47 | 296 4.85 | 12 .25 | 9 .25 | .4 .02 | 2.1 .03 | 281 | 246 | 242 | 4 |
| 59 | SW SW sec. 33 | 190.5 | Cheyenne | Sept. 20 | 61 | 5.8 | 256 12.77 | 110 9.04 | 3,068 133.48 | 128 2.10 | 1,141 23.73 | 4,588 129.38 | 1.3 .07 | .75 .01 | 9235 | 1090 | 105 | 985 |
| 60 | NE SE sec. 34 | 59.4 | Meade | Sept. 19 | 61 | .47 | 82 4.09 | 20 1.64 | 45 1.95 | 309e 5.07 | 38 .79 | 61 1.72 | .4 .02 | 4.9 .08 | 406 | 286 | 254 | 32 |
| T. 29 S., R. 18 W. | ||||||||||||||||||
| 61 | NW NW sec. 7 | 180.5 | Ogallala | Oct. 1 | 60 | .64 | 68 3.39 | 12 .99 | 14 .61 | 265 4.35 | 12 .25 | 8.2 .23 | .3 .02 | 8.8 .14 | 256 | 219 | 218 | 1 |
| 65 | SE NE sec. 30 | 94.5 | Meade and/or Ogallala | Sept. 19 | 60 | 2.7 | 120 5.99 | 10 .82 | 5.3 .23 | 370 6.07 | 11 .23 | 6.5 .18 | .1 .01 | 34 .55 | 375 | 340 | 304 | 36 |
| 66 | NW SE sec. 35 | 275 | Cheyenne | Sept. 20 | 62 | 8.5 | 70 3.49 | 38 3.12 | 1,075 46.75 | 215 3.53 | 277 5.76 | 1,560 43.99 | 1.2 .06 | 1 .02 | 3138 | 330 | 176 | 154 |
| T. 30 S., R. 16 W. | ||||||||||||||||||
| 75 | NW SW sec. 3 | 18 | Recent (alluvium) | Sept. 20 | 66 | .14 | 91 4.54 | 25 2.06 | 74 3.23 | 345 5.66 | 128 2.66 | 50 1.41 | .7 .04 | 3.8 .06 | 545 | 330 | 283 | 47 |
| 76 | SW NW sec. 9 | 30 | Recent (alluvium) | Oct. 1 | 60 | 1.0 | 520 25.95 | 118 9.7 | 59 2.56 | 428 7.02 | 1,454 30.24 | 31 .87 | 1.2 .06 | 1.1 .02 | 2400 | 1782 | 351 | 1431 |
| 82 | NE SW sec. 23 | 55 | Whitehorse | Sept. 20 | 62 | 22 | 321 16.02 | 83 6.82 | 80 3.50 | 213 3.49 | 1,032 21.47 | 47 1.32 | .8 .04 | 1.3 .02 | 1694 | 1142 | 174 | 968 |
| T. 30 S., R. 17 W. | ||||||||||||||||||
| 84 | SW SW sec. 20 | 36 | Meade | Sept. 20 | 60 | 0 | 57 2.84 | 11 .9 | 14 .63 | 219 3.59 | 9.5 .2 | 9.2 .26 | .6 .03 | 18 .29 | 229 | 187 | 180 | 7 |
| 86 | SE SW sec. 34 | 96 | Meade | Sept. 20 | 60 | .22 | 63 3.14 | 10 .82 | 16 .71 | 262 4.30 | 6 .12 | 7.8 .22 | .3 .02 | .89 .01 | 235 | 198 | 198f | 0 |
| T. 30 S., R. 18 W. | ||||||||||||||||||
| 87 | NW SW sec. 1 | 24 | Meade | Sept. 20 | 60 | .22 | 70 3.49 | 16 1.32 | 23 1.01 | 318 5.22 | 2.2 .04 | 18 .51 | .4 .02 | 1.9 .03 | 291 | 240 | 240g | 0 |
| 88 | NW NW sec. 3 | 18 | Recent (alluvium) | Sept. 20 | 62 | .29 | 100 4.99 | 16 1.32 | 42 1.85 | 388 6.36 | 37 .77 | 28 .79 | .3 .02 | 14 .22 | 432 | 316 | 316h | 0 |
| 90 | NW NW sec. 20 | 59.3 | Meade | Sept. 20 | 59 | .36 | 113 5.64 | 7.5 .62 | 5.7 .25 | 333 5.46 | 9.6 .2 | 6.5 .18 | .1 .01 | 41 .66 | 350 | 313 | 273 | 40 |
| T. 30 S., R. 20 W. | ||||||||||||||||||
| 97 | NE NE sec. 2 | 94.5 | Meade | Sept. 19 | 60 | .04 | 61 3.04 | 17 1.4 | 14 .59 | 270 4.43 | 11 .23 | 10 .28 | .4 .02 | 4.4 .07 | 253 | 222 | 222 | 0 |
| a. One part per million is equivalent to one pound of substance per million pounds of water or 8.33 pounds per million gallons of water. b. An equivalent per million (e.p.m.) is a unit chemical equivalent weight of solute per million unit weights of solution. Concentration in equivalents per million is calculated by dividing concentration in parts per million by the chemical combining weight of the substance or ion. c. Calculated. d. Includes 7.2 parts per million of carbonate (CO3). e. Includes 3.6 parts per million of carbonate (CO3). f. Total alkalinity, 215 parts per million; excess alkalinity, 17 parts per million. g. Total alkalinity, 261 parts per million; excess alkalinity, 21 parts per million. h. Total alkalinity, 318 parts per million; excess alkalinity, 2 parts per million. |
||||||||||||||||||
Table 5--Analyses of waters from typical springs in Kiowa County, Kansas. Analyzed by H. A. Stoltenberg. Parts per milliona and equivalents per millionb (in italics).
| No. on Fig. 8 |
Location | Geologic subdivision | Date of collection, 1941 |
Temp. (F°) |
Iron (Fe) |
Calcium (Ca) |
Magnesium (Mg) |
Sodium and potassium (Na+K)c |
Bicar- bonate (HCO3) |
Sulfate (SO4) |
Chloride (Cl) |
Fluoride (Fl) |
Nitrate (NO3) |
Dissolved solids |
Hardness (calculated as CaCO3) |
||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total | Carbonate | Non- carbonate |
|||||||||||||||
| T. 29 S., R. 16 W. | |||||||||||||||||
| 6 | SW SE sec. 20 | Meade | Oct. 1 | 60 | 0.26 | 83 4.14 | 9 .74 | 20 .89 | 315 5.17 | 14 .29 | 10 .28 | 0.4 .02 | 0.58 .01 | 295 | 244 | 244d | 0 |
| 10 | NE SE sec. 23 | Meade | Sept. 19 | 64 | .04 | 58 2.89 | 8 .66 | 14 .61 | 216 3.54 | 9.4 .2 | 9.8 .28 | .3 .02 | 7.5 .12 | 215 | 178 | 177 | 1 |
| T. 29 S., R. 17 W. | |||||||||||||||||
| 13 | SE SW sec. 10 | Meade | Sept. 20 | 60 | 0 | 67 3.34 | 9.6 .79 | 9.7 .42 | 248 4.07 | 7.2 .15 | 7 .2 | .2 .01 | 7.5 .12 | 232 | 206 | 204 | 2 |
| T. 30 S., R. 18 W. | |||||||||||||||||
| 16 | SW SE sec. 2 | Kiowa | Oct. 1 | 60 | .06 | 61 3.04 | 14 1.15 | 37 1.59 | 273 4.48 | 21 .44 | 28 .79 | .6 .03 | 2.4 .04 | 300 | 210 | 210e | 0 |
| 18 | NW SW sec. 11 | Kiowa | Sept. 20 | 66 | .6 | 48 2.40 | 9.8 .8 | 15 .64 | 207 3.39 | 13 .27 | 4.2 .12 | .9 .05 | .84 .01 | 196 | 160 | 160f | 0 |
| a. One part per million is equivalent to one pound of substance per million pounds of water or 8.33 pounds per million gallons of water. b. An equivalent per million (e.p.m.) is a unit chemical equivalent weight of solute per million unit weights of solution. Concentration in equivalents per million is calculated by dividing concentration in parts per million by the chemical combining weight of the substance or ion. c. Calculated. d. Total alkalinity, 258 parts per million, excess alkalinity, 14 parts per million. e. Total alkalinity, 224 parts per million, excess alkalinity, 14 parts per million. f. Total alkalinity, 170 parts per million; excess alkalinity, 10 parts per million. |
|||||||||||||||||
Chemical Constituents in Relation to Use
The following discussion of the chemical constituents of ground water has been adapted from publications of the United States Geological Survey.
Dissolved solids
The residue left after a natural water has evaporated consists of rock materials which may include some organic material and some water of crystallization. Waters containing less than 500 parts per million of dissolved solids are generally entirely satisfactory for domestic use, except for the difficulties resulting from their hardness and, in some areas, excessive iron corrosiveness. Waters having more than 1,000 parts per million are as a rule not satisfactory, for they are likely to contain enough of certain constituents to produce a noticeable taste or to make the water unsuitable in some other respects.
The ground waters from more than half of the wells and all of the springs sampled in Kiowa County contained less than 300 parts per million of dissolved solids, and are entirely satisfactory for most ordinary purposes. The waters from five of the wells sampled (60, 65, 75, 88, and 90) contained between 300 and 550 parts per million of dissolved solids, the waters from four wells (55, 66, 76, and 82) contained between 1,000 and 5,000 parts, and the water from well 59 contained 9,235 parts.
Hardness
The hardness of water, which is the property that generally receives the most attention, is most commonly recognized by its effects when soap is used with the water. Calcium and magnesium cause virtually all the hardness of ordinary waters. These constituents are also the active agents in the formation of the greater part of the scale in steam boilers and in other vessels in which water is heated or evaporated.
In addition to the total hardness the table of analyses shows the carbonate hardness and the noncarbonate hardness. The carbonate hardness is that due to the presence of calcium and magnesium bicarbonates. It is almost completely removed by boiling. In some reports this type of hardness is called temporary hardness. The noncarbonate hardness is due to the presence of sulfates or chlorides of calcium and magnesium, but it cannot be removed by boiling and has sometimes been called permanent hardness. With reference to use with soaps there is no difference between the carbonate and noncarbonate hardness. In general the noncarbonate hardness forms harder scale in steam boilers.
Water having a hardness of less than 50 parts per million is generally rated as soft, and its treatment for removal of hardness under ordinary circumstances is not necessary. Hardness between 50 and 150 parts per million does not seriously interfere with the use of water for most purposes, but it does slightly increase the consumption of soap, and its removal by a softening process is profitable for laundries or other industries using large quantities of soap. Waters in the upper part of this range of hardness will cause considerable scale in steam boilers. Hardness of more than 150 parts per million can be noticed by anyone, and if the hardness is 200 or 300 parts per million it is common practice to soften water for household use or to install cisterns to collect soft rain water. Where municipal water supplies are softened, an attempt is generally made to reduce the hardness to 50 or 80 parts per million. The additional improvement from further softening of a whole public supply is not deemed worth the increase in cost.
Water samples collected in Kiowa County ranged in hardness from 86 to 1,782 parts per million. Twelve of the samples of water had less than 200 parts per million of hardness, eleven had between 201 and 300 parts, six had between 301 and 400 parts, and three (59, 76, and 82) had more than 500 parts. The water in neither of the two public water supplies in the county is treated, but it is necessary to treat the industrial supplies to reduce the hardness.
Iron
Next to hardness, iron is the constituent of natural waters that in general receives the most attention. The quantity of iron in ground waters may differ greatly from place to place, even though the waters are derived from the same formation. If a water contains much more than 0.1 part per million of iron the excess may separate out and settle as a reddish sediment. Iron, which may be present in sufficient quantity to give a disagreeable taste and to stain cooking utensils, may be removed from most waters by simple aeration and filtration, but a few waters require the addition of lime or some other substance.
Of the 32 samples of water analyzed from Kiowa County, nine contained less than 0.1 part per million of iron, 15 contained between 0.1 and 1.0 part, four (wells 27, 44, 49, and 65) contained between 1.1 and 5 parts, and four (wells 58, 59, 66, and 82) contained more than 5 parts.
Chloride
Chloride is in abundant constituent of sea water. It is dissolved in small quantities from rock materials and in some localities comes from sewage. The sources of chloride are many, however, and its presence in large quantities cannot be taken as a definite indication of pollution. Chloride has little effect on the suitability of water for ordinary use unless there is enough to give the taste of salt. Waters high in chloride may be corrosive when used in steam boilers.
All of the samples of water collected in Kiowa County had a low chloride content, except those from wells 55, 59, and 66, which contained 1,210, 4,5S8, and 1,560 parts per million of chloride, respectively.
Fluoride
Although determinable quantities of fluoride are not so common as fairly large quantities of the other constituents of natural waters, it is desirable to know the amount of fluoride present in waters that are likely to be used by children. Fluoride in water has been shown to be associated with the dental defect known as mottled enamel, which may appear on the teeth of children who drink water containing fluoride during the period when their permanent teeth are formed. It has been said that waters containing 1 part per million or more of fluoride are likely to produce mottled enamel, although the effect of 1 part per million is not usually very serious (Dean, 1936, pp. 1269-1272). If the water contains as much 4 parts per million of fluoride, 90 percent of the children who drink it are likely to have teeth with mottled enamel, and 35 percent or more of these cases will be classified as moderate or worse. Small quantities of fluoride, not, sufficient to cause mottled enamel, are likely to be beneficial by decreasing dental caries (Dean, Arnold, and Elvove, 1942, pp,. 1155-1179).
The fluoide content of the water samples from Kiowa County was low. Of the 32 samples analyzed, 28 contained less than 1 part per million of fluoride and four (55, 59, 66, and 76) contained between 1 and 2 parts.
Water for irrigation
The suitability of water for use in irrigation is commonly thought to depend mainly on the total quantity of soluble salts the ratio of the quantity of sodium to the total quantity of sodium, calcium, and magnesium together. The quantity of chloride may be large enough to affect the use of the water, and in some areas other constituents, such as boron, may be present in sufficient quantity to cause difficulty. With reference to the quality of irrigation waters in southern California, Scofield (1933) suggests that, if the total concentration of dissolved salts is less than 700 parts per million there is not, much probability of harmful effects in irrigation use, but if it exceeds 2,000 parts per million there is a strong probability of damage to either the crops or the land, or both. Water containing less than 50 percent sodium (the percentage being calculated as 100 times the ratio of the total bases, in equivalents) is not likely to be injurious, but if it contains more than 60 percent, its use is inadvisable. A chloride content of less than 142 parts per million is not objectionable, but more than 355 parts per million is undesirable. Similarly, a sulfate content of less than 192 parts per milllion is not objectionable, but more than 480 parts per million may be injurious. The harmfulness of these various salts in irrigation water is so dependent on the nature of the land and crops, the manner of use, and drainage that no hard and fast limits can be adopted.
Five of the samples (55, 59, 66, 76, and 82 collected in Kiowa County had mineral concentrations in excess of the safe limits given by Scofield and therefore are unsuitable for irrigation use. The concentration of dissolved solids, sulfite, and chloride, and percent sodium for each of these samples are given in Table 6. All but one (76) of these samples were obtained from wells that, draw water from consolidated deposits in areas where irrigation is not feasible. The water in well 76 comes from alluvium and is used in an air-conditioning unit.
Table 6--Concentration of dissolved solids, sulphate, and chloride, and percent sodium in five samples of water collected from wells in Kiowa County, Kansas. Percent sodium = (100 Na) / (Na + Ca + Mg), in which the three chemical constituents are expressed in equivalents per million.
| Well No. |
Dissolved solids (parts per million) |
Sulphate (SO4) parts per million |
Chloride (Cl) parts per million |
Sodium (percent) |
|---|---|---|---|---|
| 55 | 4,692 | 1,722 | 1,210 | 91.6 |
| 59 | 9,235 | 1,141 | 4,588 | 85.9 |
| 66 | 3,138 | 277 | 1,560 | 87.6 |
| 76 | 2,400 | 1,454 | 31 | 6.7 |
| 82 | 1,694 | 1,032 | 47 | 13.3 |
Sanitary Considerations
Every precaution should be used to protect domestic and public water supplies from pollution by organic material. A large percentage of the population of Kiowa County is dependent on private water supplies from wells, and it rests chiefly with the drillers and individual well-owners to observe precautions to insure a safe and wholesome water supply. It is obvious that a well should not be located where there are possible sources of pollution nor where surface water can descend directly to the water table. Drainage from cesspools and privies is particularly dangerous. Every well should be so constructed as to seal off all surface water. As a general rule dug wells are more subject to contamination from surface water than are drilled wells, mainly because they are generally not effectively sealed at the surface.
Relation to Stratigraphy
Samples of water were collected from wells and springs that are supplied by the following water-bearing formations: Flowerpot shale and Whitehorse sandstone (Permian), Cheyenne sandstone and Kiowa shale (Cretaceous), Ogallala and Meade formations (Pliocene and Pleistocene), and alluvium (Recent). The quality of the water from each formation is shown graphically in Figure 7 and is discussed in more detail by formations under water-bearing characteristics of rock formations. Since the quality of water in the Meade and Ogallala formations is similar and since it is difficult to determine from which sediments some wells obtain water, it seems best to discuss the waters of the two formations as a unit. Moreover, some wells obtain water from both formations.
Fig. 7--Analyses of water from the principal water-bearing formations in Kiowa County.
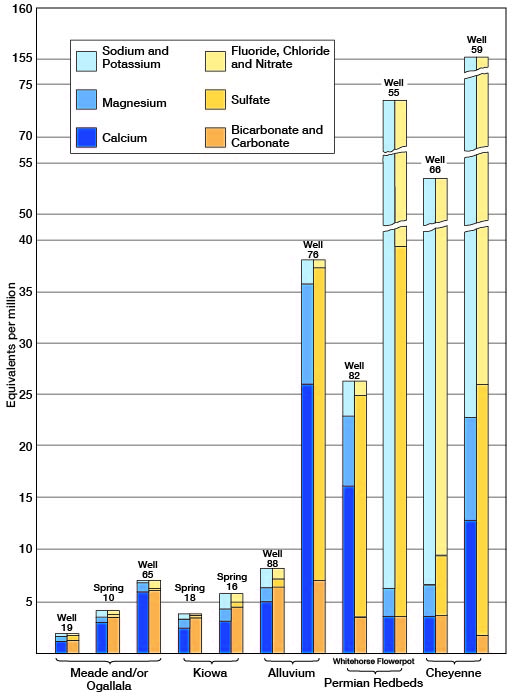
The samples of water from the different water-bearing formations differed widely in their content of dissolved mineral matter. The lowest concentrations of mineral matter were found in the waters from the Meade and Ogallala formations. The two samples of water from the Kiowa shale fall in the middle range of concentration of the waters from the Meade and Ogallala. Waters from the Permian redbeds and the Cheyenne sandstone had the greatest concentrations of dissolved mineral matter and are in general the poorest waters found in the county. The concentration of the three samples from alluvium differed greatly. Two of the samples were only slightly more concentrated than the poorest waters from the Meade and Ogallala formations, but one sample was as highly mineralized as one sample from the Permian.
The difference in the amount of mineral concentration in the two waters from the Permian redbeds is due to the difference in the mineral content of the deposits from which they came. The Flowerpot shale consists predominantly of gypsiferous shales but in some places contains beds of gypsum, gypsiferous sandstone, or rock salt (Norton, 1939, p. 1792) ; it yields mineralized water that is high in sodium, chloride, and sulfate (analysis 55). The Whitehorse consists almost entirely of iron-cemented fine-grained sandstone and yields very hard water that is high in sulfate and iron but relatively low in sodium and chloride (analysis 82).
Prev Page--Recovery, Utilization || Next Page--Formations
Kansas Geological Survey, Geology
Placed on web Feb. 4, 2008; originally published Feb. 1948.
Comments to webadmin@kgs.ku.edu
The URL for this page is http://www.kgs.ku.edu/General/Geology/Kiowa/05_gw4.html